| click here to download brochure |

|

|

The AC6000 series Glycated Hemoglobin Analyzer was launched in 2010. The AC6601 model is the first LPLC method HbA1c analytical device in China to obtain a medical device registration certificate.

The product has undergone 15 years of stable operation. Global installations have exceeded 4,000 units, serving more than 30 countries and thousands of medical institutions.

Relying on the company’s core patented technology, the equipment coefficient of variation (CV) ≤3.0%, supports continuous batch testing, and its performance is significantly superior to conventional POCT products; it provides a fully automatic sampling system, is easy to operate with low maintenance cost, can seamlessly connect with HIS/LIS systems, and perfectly adapts to primary and secondary hospital scenarios.
The product is equipped with original factory calibrators and quality control materials, adopts a metrological traceability system, supports both venous whole blood and fingertip capillary blood testing, has passed multiple international certifications including CE, NGSP, ISO13485, ISO9001, National Medical Products Administration, and has obtained medical device registration qualifications in multiple countries.
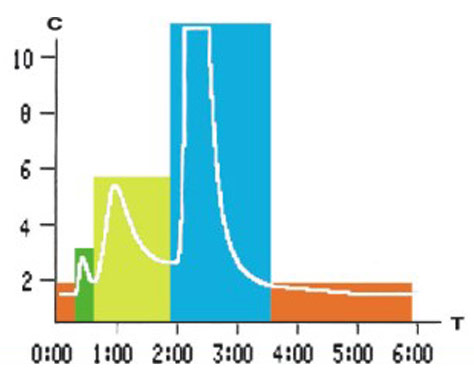
● Classic methodological principle — ion-exchange liquid chromatography.
● Core patented technology HbA1c four-gradient elution method, separating glycated hemoglobin.
● Micro-volume whole blood, applicable for laboratory and clinical outpatient use.

● High-performance liquid chromatography column, efficient chromatographic column for more than 330 tests.

● High-sensitivity 415nm LED integrated photometer.
● Internal preheating, precise temperature control.
● Constant temperature device, improving repeatability and accuracy of test results.
● Real-time display of analytical chromatogram, making precision visible.

● AC6601 Glycated Hemoglobin Analyzer reagents: Eluent A, Eluent B, Eluent C, Eluent D, Hemolysis reagent.
| Supporting reagent product name | Glycated Hemoglobin Testing Reagent (Liquid Chromatography Method). |
| Repeatability | CV≤3.0% (International standard). |
| Reagent configuration | Each standard kit can test more than 330 samples. |
| Batch analysis | The test report simultaneously outputs IFCC concentration value, NGSP area percentage, and ADAG average blood glucose. |
| Power supply | ~220V±22V、50Hz±1Hz、215VA |